Seminar: “A Uniform-Shear-Rate Microfluidic Bioreactor for Real-Time Analysis of Proplatelet Formation and Rapidly-Released Platelets”
21 Jun 17 iBET-ITQB Room 2.13
ABSTRACT:
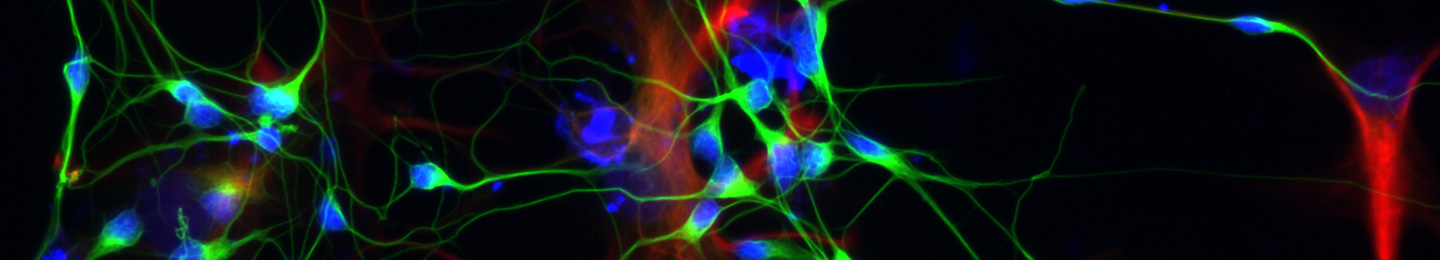
Platelet transfusions are entirely dependent on human volunteer donors, and are limited by platelet storage at room temperature, a 5-day platelet shelf life, and differences in donor/recipient histocompatibility. These challenges invite the opportunity to generate platelets ex vivo. Much progress has been made in generating large numbers of culture-derived megakaryocytes (Mks, the precursor cells to platelets), but stimulating a high percentage of Mks to undergo terminal maturation and platelet release in vitro remains a major challenge. In vivo, platelets are formed when bone marrow Mks extend long, cytoplasmic projections, called proplatelets (proPLTs), into the sinusoid where shear forces accelerate proPLT elongation and release platelets into circulation. Platelets may also be released directly by Mks in lung capillaries. Recent studies have demonstrated the utility of shear forces to enhance platelet release from Mks in vitro. We are exploring the production of platelet-like particles (PLPs) within a microfluidic bioreactor that utilizes shear forces on Mks to generate proPLTs and PLPs. We performed computational fluid dynamics (CFD) analysis of several published platelet microbioreactor systems, and used the results to develop a new bioreactor – with well-defined flow patterns and uniform shear profiles. Experimental studies were conducted to validate the simulations in terms of streamline profiles and flow patterns – with and without cell capture. Furthermore, the bioreactor supports a wide physiological shear rate range and fits within the stage of a fluorescent microscope housed in an incubator for real-time analysis of proPLT formation and PLP release. The videos and images captured within our system show that the new bioreactor not only promotes the prototypical proPLT formation process with beads-on-a-string morphology, but also supports rapid release of individual PLPs – which has been observed in vivo, but not previously reported for platelet bioreactors. Furthermore, we showed that modulating shear forces and flow patterns real-time within the system had an immediate and significant impact to proPLT and PLP generation. By identifying positive operating conditions within a physiologically relevant environment, this new bioreactor will be a useful tool for the study and analysis of proPLT/PLP formation that will further understanding of ex vivo platelet release. The system can be further scaled, for example, through parallelization of reactors. We previously showed that PLPs can be efficiently separated from Mks using spinning-membrane filtration.
Professor William M. Miller short Bio
Bill received a BS in chemical engineering from Lehigh University and an MS from MIT. After 8 years with Rohm & Haas and Air Products & Chemicals, he obtained a PhD from the University of California, Berkeley. Bill has been at Northwestern since 1987; he served as Chemical and Biological Engineering department chair and currently directs the MS in Biotechnology Program and the NIH predoctoral Biotechnology Training Program. Bill received a Presidential Young Investigator Award from NSF; chaired the American Institute of Chemical Engineers Food, Pharmaceutical and Bioengineering Division; served on the Scientific Advisory Boards of the Australian Stem Cell Centre and the Stem Cell Network of Canada; and was a member of the National Research Council Task Group to evaluate NASA's Biotechnology Facility for the International Space Station. He is a Fellow of the American Association for the Advancement of Science and the American Institute of Medical and Biological Engineers. Bill has authored more than 110 peer-reviewed publications and supervised 36 PhD students. He was Editor of the Biochemical Engineering Journal for 9 years and serves on editorial boards for Biotechnology Progress, Biotechnology & Bioengineering, and the Biotechnology Journal.